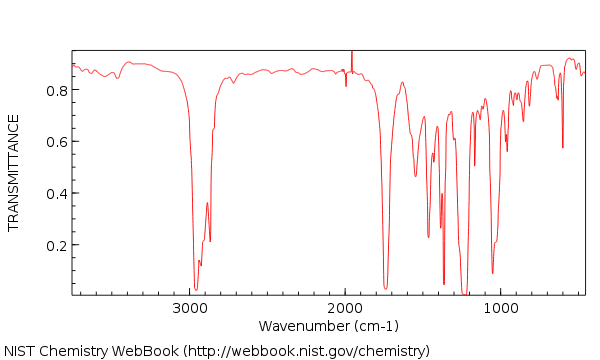
The calculation method and formulation are kept as simple as possible to avoid degrading the performance of numerical models of water-CO 2 flows for which they are intended. (1992), covering a broader range of temperatures and experimental data than those authors, and is readily expandable to a nonideal liquid phase. The procedure is an extension of that used by King et al. In this paper, a noniterative procedure is presented to calculate the composition of the compressed CO 2 and liquid H 2 O phases at equilibrium, based on equating chemical potentials and using the Redlich-Kwong equation of state to express departure from ideal behavior. For the most part, mutual solubilities reported from various sources are in good agreement. These data cover the two-phase region where a CO 2 -rich phase (generally gas) and an H 2 O-rich liquid coexist and are reported as the mutual solubilities of H 2 O and CO 2 in the two coexisting phases. For this purpose, published experimental P - T - X data in this temperature and pressure range are reviewed. read more read lessĪbstract: Evaluating the feasibility of CO 2 geologic sequestration requires the use of pressure-temperature-composition ( P - T - X ) data for mixtures of CO 2 and H 2 O at moderate pressures and temperatures (typically below 500 bar and below 100☌). On the Applicability of DFT to Anions and theFuture of DFT EA Predictions251D. Theoretical EAs for Species with UnknownExperimental EAs251C. Statistical Analysis of DFT Results ThroughComparisons to Experiment and OtherTheoretical Methods248B. Details of Density Functional MethodsEmployed in Tables IV. Layout of Tables 8 and 9: Theoretical DFTElectron Affinities247F. Density Functional Theory (DFT) andElectron Affinities245E. 
Basis Sets and Theoretical Electron Affinities 244D. Present Status of Theoretical Electron AffinityPredictions243C. Theoretical Predictions of MolecularElectron Affinities243B. Theoretical Predictions of Atomic ElectronAffinities2422. Theoretical Determination of Electron Affinities 242A. Layout of Table 10: ExperimentalPhotoelectron Electron Affinities242III. Some Thermochemical Uses of ElectronAffinities241F. Time-of-Flight Negative Ion PhotoelectronSpectroscopy239E. Experimental Photoelectron Electron Affinities 235A. 
Definitions of Molecular Electron Affinities 233II. Definitions of Atomic Electron Affinities 233B. But at least using a template instead of just using a URL ensures that the links from Wikipedia are centralized and hopefully easier to fix.Abstract: I. Note: There is always a risk that NIST may change the structure of the website and the query parameters. So a way of including just about everything would be to use a mask value of FFFF. The current meaning of this field is a hexadecimal number resulting from the sum of the following bits: 1 Gas phase thermochemical dataĨ00 Vibrational and/or electronic energy levels mask this can be obtained from the URL and determines which sort of data is displayed. units either CAL (for calorie-based units) or SI. accessdate this can be useful because the WebBook could change as data is updated. If not present, the ID will be used as a name. name the name of the substance, used for the link name. (eds.) NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2008) id the NIST id for the substance (can be obtained from the URL). An example using all available parameters is the following:Īmmonium chloride in Linstrom, Peter J. To link to a specific substance, the id parameter needs to be supplied. (eds.) NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) By default, the template produces a generic citation to the WebBook: This template produces a citation to the NIST Chemistry WebBook.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
